By Picasso Vasquez, Genetics and Genomics ‘20
Author’s Note: One of the main goals for my upper division UWP class was to write about a recent scientific discovery. I decided to write about CAR-T cell therapy because this summer I interned at a pharmaceutical company and worked on a project that involved using machine learning to optimize the CAR-T manufacturing process. I think readers would benefit from this article because it talks about a recent development in cancer therapy.
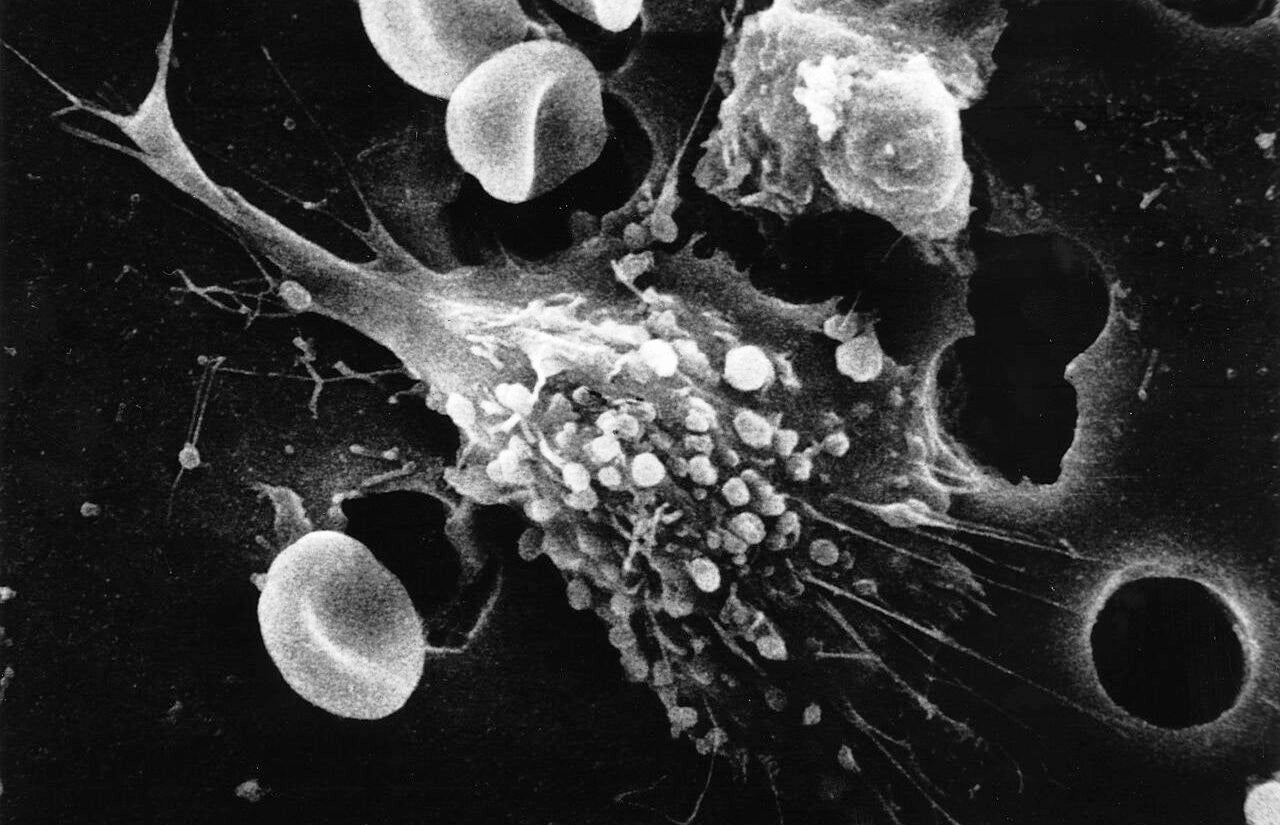
“There’s no precedent for this in cancer medicine.” Dr. Carl June is the director of the Center for Cellular Immunotherapies and the director of the Parker Institute for Cancer Immunotherapy at the University of Pennsylvania. June and his colleagues were the first to use CAR-T, which has since revolutionized personal cancer immunotherapy [1]. “They were like modern-day Lazarus cases,” said Dr. June, referencing the resurrection of Saint Lazarus in the Gospel of John and how it parallels the first two patients to receive CAR-T. CAR-T, or chimeric antigen receptor T-cell, is a novel cancer immunotherapy that uses a person’s own immune system to fight off cancerous cells existing within their body [1].
Last summer, I had the opportunity to venture across the country from Davis, California, to Springhouse, Pennsylvania, where I worked for 12 weeks as a computational biologist. One of the projects I worked on was using machine learning models to improve upon the manufacturing process of CAR-T, with the goal of reducing the cost of the therapy. The manufacturing process begins when T-cells are collected from the hospitalized patient through a process called leukapheresis. In this process, the T-cells are frozen and shipped to the manufacturing facility, such as the one I worked at this summer, where they are then grown up in large bioreactors. On day three, the T-cells are genetically engineered to be selective towards the patient’s cancer by the addition of the chimeric antigen receptor; this process turns the T-cells into CAR-T cells [2]. For the next seven days, the bioengineered T-cells continue to grow and multiply in the bioreactor. On day 10, the T-cells are frozen and shipped back to the hospital where they are injected back into the patient. Over the 10 days prior to receiving the CAR-T cells, the patient is given chemotherapy to prepare their body for inoculation of the immunotherapy [2]. This whole process is very expensive and as Dr. June put it in his TedMed talk, “it can cost up to 150,000 dollars to make the CAR-T cells for each patient.” But the cost does not stop there; when you include the cost of treating other complications, the cost “can reach one million dollars per patient” [1].
The biggest problem with fighting cancer is that cancer cells are the result of normal cells in your body gone wrong. Because cancer cells look so similar to the normal cells, the human body’s natural immune system, which consists of B and T-cells, is unable to discern the difference between them and will be unable to fight off the cancer. The concept underlying CAR-T is to isolate a patient’s T-cells and genetically engineer them to express a protein, called a receptor, that can directly recognize and target the cancer cells [2]. The inclusion of the genetically modified receptor allows the newly created CAR-T cells to bind cancer cells by finding the conjugate antigen to the newly added receptor. Once the bond between receptor and antigen has been formed, the CAR-T cells become cytotoxic and release small molecules that signal the cancer cell to begin apoptosis [3]. Although there has always been drugs that help your body’s T-cells fight cancer, CAR-T breaks the mold by showing great efficacy and selectivity. Dr. June stated “27 out of 30 patients, the first 30 we treated, or 90 percent, had a complete remission after CAR-T cells.” He then goes on to say, “companies often declare success in a cancer trial if 15 percent of the patients had a complete response rate” [1].
As amazing as the results of CAR-T have been, this wonderful success did not happen overnight. According to Dr. June, “CAR T-cell therapies came to us after a 30-year journey, along with a road full of setbacks and surprises.” One of these setbacks is the side effects that result from the delivery of CAR-T cells. When T-cells find their corresponding antigen, in this case the receptor on the cancer cells, they begin to multiply and proliferate at very high levels. For patients who have received the therapy, this is a good sign because the increase in T-cells indicates that the therapy is working. When T-cells rapidly proliferate, they produce molecules called cytokines. Cytokines are small signaling proteins that guide other cells around them on what to do. During CAR-T, the T cells rapidly produce a cytokine called IL-6, or interleukin-6, which induces inflammation, fever, and even organ failure when produced in high amounts [3].
According to Dr. June, the first patient to receive CAR-T had “weeks to live and … already paid for his funeral.” When he was infused with CAR-T, the patient had a high fever and fell comatose for 28 days [1]. When he awoke from his coma, he was examined by doctors and they found that his leukemia had been completely eliminated from his body, meaning that CAR-T had worked. Dr. June reported that “the CAR-T cells had attacked the leukemia … and had dissolved between 2.9 and 7.7 pounds of tumor” [1].
Although the first patients had outstanding success, the doctors still did not know what caused the fevers and organ failures. It was not until the first child to receive CAR-T went through the treatment did they discover the cause of the adverse reaction. Emily Whitehead, at six years old, was the first child to be enrolled in the CAR-T clinical trial [1]. Emily was diagnosed with acute lymphoblastic leukemia (ALL), an advanced, incurable form of leukemia. After she received the infusion of CAR-T, she experienced the same symptoms of the prior patient. “By day three, she was comatose and on life support for kidney failure, lung failure, and coma. Her fever was as high as 106 degrees Fahrenheit for three days. And we didn’t know what was causing those fevers” [1]. While running tests on Emily, the doctors found that there was an upregulation of IL-6 in her blood. Dr. June suggested that they administer Tocilizumab to combat increased IL-6 levels. After contacting Emily’s parents and the review board, Emily was given Tocilizumab and “Within hours after treatment with Tocilizumab, Emily began to improve very rapidly. Twenty-three days after her treatment, she was declared cancer-free. And today, she’s 12 years old and still in remission” [1]. Currently, two versions of CAR-T have been approved by the FDA, Yescarta and Kymriah, which treat diffuse large B-cell lymphoma (DLBCL) and acute lymphoblastic leukemia (ALL) respectively [1].
The whole process is very stressful and time sensitive. This long manufacturing task results in the million-dollar price tag on CAR-T and is why only patients in the worst medical states can receive CAR-T [1]. However, as Dr. June states, “the cost of failure is even worse.” Despite the financial cost and difficult manufacturing process, CAR-T has elevated cancer therapy to a new level and set a new standard of care. However, there is still much work to be done. The current CAR-T drugs have only been shown to be effective against liquid based cancers such as lymphomas and non-effective against solid tumor cancers [4]. Regardless, research into improving the process of CAR-T continues to be done both at the academic level and the industrial level.
References:
- June, Carl. “A ‘living drug’ that could change the way we treat cancer.” TEDMED, Nov. 2018, ted.com/talks/carl_june_a_living_drug_that_could_change_the_way_we_treat_cancer.
- Tyagarajan S, Spencer T, Smith J. 2019. Optimizing CAR-T Cell Manufacturing Processes during Pivotal Clinical Trials. Mol Ther. 16: 136-144.
- Maude SL, Laetch TW, Buechner J, et al. 2018. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N Engl J Med. 378: 439-448.
- O’Rourke DM, Nasrallah MP, Desai A, et al. 2017. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med. 9: 399.