By Jessica Lee, Biochemistry and Molecular Biology ‘21
Author’s Note: Alarmed by the fact that so many Americans are skeptical of receiving a COVID-19 vaccine, I wanted to write an article delving into the reasons why public trust in medical institutions has waned. I look to previous breaches of trust to propose public health messaging strategies for the rollout of the highly anticipated COVID-19 vaccine.
As of November 2020, approximately 63% of Americans say they would not be willing to immediately receive a COVID-19 vaccine—even if the vaccine was approved by the Food and Drug Administration (FDA) and free of cost [1]. Public willingness to receive a COVID-19 vaccine has rebounded since its all time low of 50% in September of 2020. The fluctuation in willingness to be vaccinated reflects how the public perceives undue influence on the vaccine development and regulation process. A successful vaccine distribution process will require broad public support to control the ongoing global pandemic.
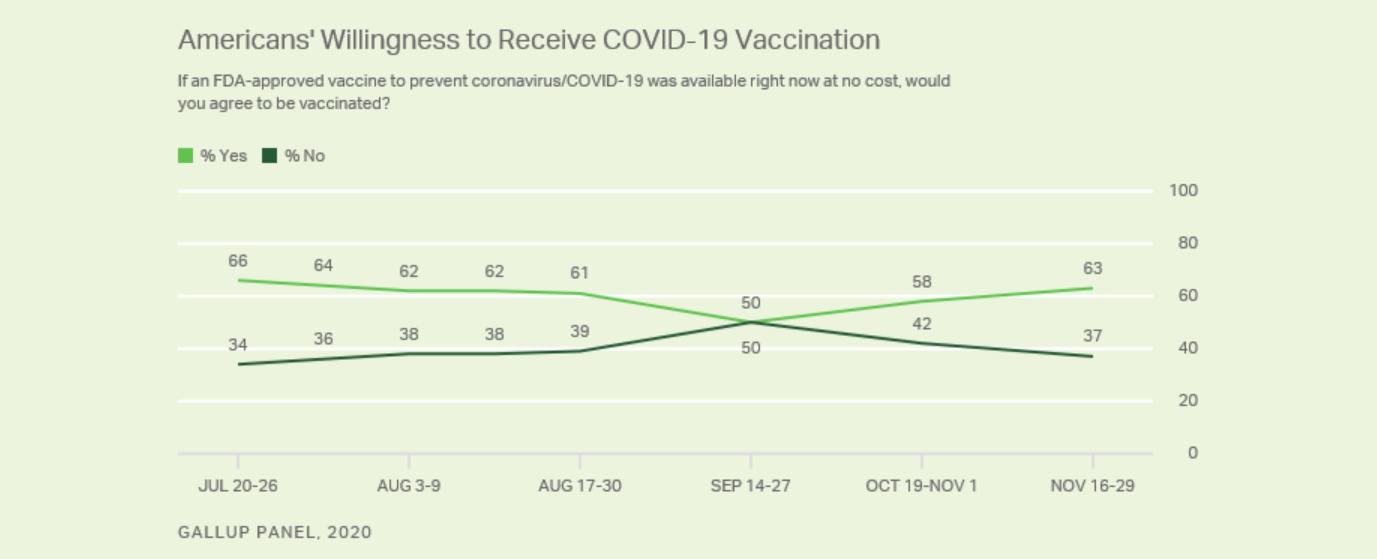
[1]
The human and economic consequences of the COVID-19 pandemic are staggering: over 350,000 people in the U.S. have died from COVID-19 and the unemployment rate remains high at 6.7% as compared to 3.5% in February 2020 [2, 3]. Given the devastating impacts of COVID-19 on Americans’ health and well-being, why are so many Americans skeptical of a vaccine with the potential to restore normalcy?
The history of American public and private biomedical institutions may provide useful context for Americans’ skepticism of a potential COVID-19 vaccine. The anti-vaccination movements, opioid crisis, and bumpy introduction of COVID-19 therapeutics have all contributed to waning trust in public health institutions. With the approval of Pfizer/BioNTech and Moderna vaccine candidates for the prevention of COVID-19, the biomedical community needs to foster trust by delivering correct and consistent messaging to the public as vaccines become available to the American public.
Trust in Biomedical Institutions
The modern anti-vaccination, or “anti-vax,” movement in the U.S. was sparked by Andrew Wakefield’s infamous paper published in The Lancet and perpetuated by outspoken celebrities, politicians, and social media groups [4]. Even though Wakefield’s claims about a causal relationship between the childhood measles, mumps, and rubella (MMR) vaccine and autism have since been thoroughly debunked by a scientific majority, the damage caused by his falsified research is evident as measles outbreaks continue to impact the U.S. Characterized by fantastical and conspiratorial thinking, the modern anti-vax movement has evolved to include a range of beliefs about vaccines. On social media platforms, misinformation about vaccines can include false safety concerns to conspiracies about social control.
However, there are also legitimate reasons to be skeptical of the pharmaceutical industry and its regulators. Mistakes driven by commercial interests have resulted in horrific public health crises. Motivated by profit, pharmaceutical companies misled the public about the safety of opioids, such as oxycontin, resulting in the liberal prescription of highly addictive and dangerous drugs. Opioid overdose is now one of the most common causes of preventable death in the U.S. [5]. Financial incentives can corrupt the scientific process, even corrupting leading medical experts.
Dr. Russell Portenoy, a pain specialist, received millions of dollars from the manufacturers of opioids while assuring the public that addiction risks were low [5]. When the addictive nature of opioids became evident, Portenoy defended his actions.
“My viewpoint is that I can have these relationships [and] they would benefit my research mission and to some extent, they can benefit my own pocketbook, without producing in me any tendency to engage in undue influence or misinformation,” said Portenoy [5].
In light of the unethical—and often illegal—behavior of pharmaceutical companies, the reaction of the American public is not entirely unreasonable. However, the waning trust in biomedical institutions is nonetheless a public health problem with clear consequences. In 2019, there were several outbreaks of measles among communities with low vaccination rates [6]. Over 1,200 cases were reported by the Center for Disease Control (CDC), which is the highest number of measles cases since 1992 [6]. It is important to highlight that overall measles vaccination rates are high throughout the country. However, outbreaks of deadly diseases can still occur when vaccination rates within a community dip below those needed for herd immunity. To eradicate a disease, outreach to fringe communities is necessary to ensure they buy into the vaccination process. Furthermore, vaccines must be made accessible to traditionally underserved communities. Within the context of the COVID-19 pandemic, this means that public health officials must reach out to those with anti-vaccination tendencies, ethnic minorities, and immigrant populations. Furthermore, the vaccine must be made widely accessible for the poorest citizens of all countries. Only then can COVID-19 be completely eradicated.
Number of Measles cases reported by year
Data from CDC.gov as of October 15, 2020
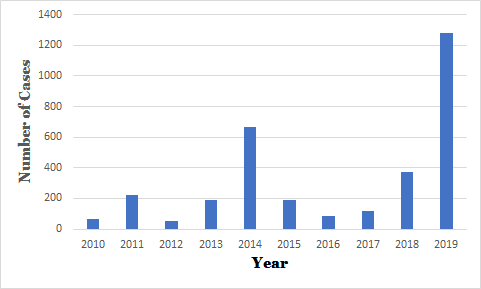
[6]
The consequences of eroded trust in biomedical institutions are even more tangible as authorities in the U.S. attempt to control the COVID-19 pandemic. Confusing, conflicting messaging and policies on cloth mask usage has resulted in a partisan gap of 16 points between Republicans and Democrats on regular mask usage [7]. Even though current data and modeling demonstrate that masks reduce infections, some Americans continue to refuse to participate in this common-sense risk reduction practice [8].
The Credibility of the Food and Drug Administration (FDA)
There has also been widespread confusion on the development of COVID-19 therapeutics. The FDA has the authority to allow the use of unapproved drugs and medical products during national emergencies under an emergency use authorization (EUA). Many COVID-19 therapeutics such as Remdesivir, hydroxychloroquine, and convalescent plasma have been granted EUAs for use in specific populations, such as hospitalized patients [9]. Importantly, medical products that are granted EUAs are not granted full FDA approval. To obtain an EUA, it must be determined that the product meets three criteria: the product may be effective in diagnosing, treating, or preventing a serious disease or condition, the known and potential benefits outweigh the risks, and there are no available alternatives [9]. Many of the EUAs granted for COVID-19 treatment have stirred controversy within the biomedical community. For instance, the FDA’s decision to grant an EUA for the use of convalescent plasma in August resulted in dissent among biomedical institutions.
A National Institutes of Health (NIH) panel rebutted the FDA’s claims by issuing this statement: “There are insufficient data for the COVID-19 Treatment Guidelines Panel to recommend either for or against the use of COVID-19 convalescent plasma for the treatment of COVID-19” [10].
Other figures in the biomedical community such as Dr. Eric Topol, the director of the Scripps Research Translational Institute, criticized the head of the FDA, Dr. Stephen Hahn, for making hyperbolic statements on the safety and efficacy of convalescent plasma and for presenting misleading data to the public [11].
“So in order to get this straight, Dr. Hahn needs to also talk to the public and say that he erred and that there is no established evidence for survival advantage of convalescent plasma. That has to be determined through randomized trials that are ongoing,” said Topol on NPR’s All Things Considered [11].
The FDA also has played a controversial part in the development of hydroxychloroquine. The agency issued an EUA for hydroxychloroquine in March only to revoke the EUA in June after adverse cardiac events were reported [12]. Whether the FDA’s actions were influenced by political pressure, corporate pressure, or a desire to save lives, the controversy around COVID-19 therapeutics degrades public trust in the FDA as an institution.
Emergency Use Authorization for Vaccines to Prevent COVID-19
The discussion on therapeutic EUAs is important since the two currently approved COVID-19 vaccines were first approved through the intermediate step of an EUA. At time of publication, Moderna and Pfizer/BioNTech have successfully completed their phase three clinical trials for COVID-19 vaccines and received EUAs from the FDA [13]. Globally, approximately twenty other vaccine candidates are also in phase three clinical trials [13]. Each clinical trial has enrolled between 30,000 and 60,000 volunteers, half of which will receive the vaccine candidate and half of which will receive a placebo [13]. Approximately 160 infections of SARS-CoV-2 will be necessary to statistically determine the efficacy of each vaccine candidate. While only 160 infections might seem small in a clinical trial of 60,000, this number allows the FDA to determine if there is a statistical significance between the two arms of the clinical trial. Interim analyses may also be conducted at fewer infections by external data safety monitoring boards [14, 15]. Such data safety monitoring boards are independent of sponsors, regulators, and the scientists conducting the clinical trials. If the external board finds statistically significant results at an interim point, then the sponsors of the clinical trial may ask the FDA to review the vaccine for an EUA [14].
Published in a non-binding guidance document, the FDA outlines the criteria for potentially obtaining an EUA for a COVID-19 vaccine. Since this guidance document is non-binding, the FDA may modify the EUA process moving forward. If a sponsor seeks an EUA at an interim analysis of a phase three clinical trial, then they must demonstrate at least 50% efficacy, have a median follow-up duration of at least two months after the administration of the last dose, and safety data that would allow the FDA to make a favorable risk-benefit analysis [16]. Furthermore, the sponsor must provide sufficient data demonstrating the ability to consistently manufacture the vaccine [16]. If the FDA believes the criteria are met for an EUA, then the vaccine candidate may be administered to certain at-risk populations while the full-approval process continues. At the time of publication, both Pfizer and Moderna have produced data from their phase three clinical trials indicating their vaccines may be over 90% effective—far surpassing the 50% efficacy threshold set by the FDA [13].
Developing and Maintaining Public Trust
The COVID-19 vaccine trials are safeguarded in many ways. The scientists at the FDA have approved the phase three clinical trial protocols and monitored phase one and two clinical trials for safety and efficacy. The oversight safety boards have watched for unexplained adverse events and paused the AstraZeneca trial when unexplained neurological symptoms presented in one participant [14]. Peer reviewers have analyzed and criticized the data and conclusions generated from phase one and two clinical trials. Furthermore, influential members of the biomedical community have spoken out when they believe mistakes have been made. Evidently, there are safety measures in place to protect the public from a dangerous or ineffective vaccine. However, safety measures are not perfect. When the FDA allowed the use of hydroxychloroquine and then revoked its EUA, the FDA weakened its authority with the general public. Even the appearance of political and commercial influence on the scientific process may elicit skepticism from the public.
How can the biomedical community increase the public’s willingness to get the COVID-19 vaccine? Certainly, consistent messaging from figures of authority is important. Furthermore, the biomedical community must continue to hold regulatory agencies, corporations, and politicians responsible for their rhetoric. There must be political, legal, or economic consequences for misleading the public and degrading trust in medical institutions. Economic consequences—for example—might range from lawsuits to executives being debarred from working in the pharmaceutical industry.
Biomedical professionals have advocated for widespread outreach to many different types of communities [15]. Social media campaigns can be effective in rapidly disseminating information by engaging users to add their own input. However, social media may also hinder outreach as demonstrated by the uncontrolled spread of misinformation by anti-vaccination groups on platforms such as Facebook [17]. Viral posts containing misinformation can seed public distrust in medical institutions. Still, polling indicates that Americans overwhelmingly trust medical professionals over industry leaders or politicians for information about vaccines [18].
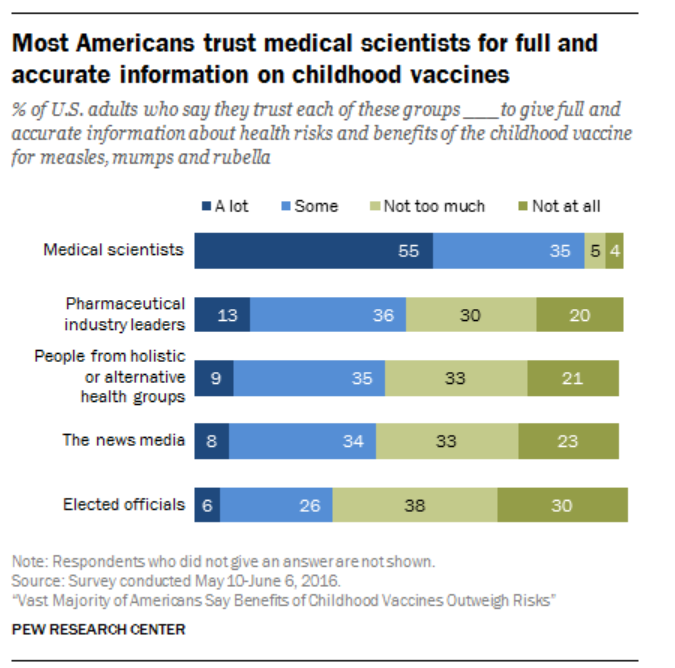
[18]
Utilizing this trust would mean elevating medical scientists as the voice communicating the state of a COVID-19 vaccine rather than relying on politicians, the news media, or industry leaders. However, it is important to communicate scientific consensus rather than relying on the voices of individual biomedical professionals. Individuals can make genuine mistakes, have differing opinions, or be corrupted; thus, it is essential that public health messaging is centered around scientific consensus.
The effectiveness of a COVID-19 vaccine on a population scale will depend on the percent of people willing to get the vaccine. To end the COVID-19 pandemic, it’s likely that most people will need to be vaccinated. To accomplish this, the biomedical community will need to work with the public to foster open and honest communication, understanding the public has relevant concerns about the influence of politics and commerce on the scientific process. By learning from previous anti-vax movements, public health professionals must counter the spread of misinformation with compelling, fact-based messaging. Ultimately, the public health community must regain the trust of the American public and appeal to Americans’ civic duty. The act of taking a vaccine is a social contract; as Dr. Topol says, “I take the vaccine to help you, not just me” [15]. The COVID-19 pandemic is one of the largest public health crises in modern times and it will require good science and good communication to solve.
References
[1] Brenan M. “Willingness to Get COVID-19 Vaccine Ticks Up to 63% in U.S.” Gallup, December, 2020.
[2] “CDC COVID Data Tracker.” Centers for Disease Control and Prevention, January 6, 2021.
[3] “Employment Situation Summary.” U.S. Bureau of Labor Statistics, December, 4, 2020.
[4] Hussain A, Ali S, Ahmed M, Hussain S. “The Anti-vaccination Movement: A Regression in Modern Medicine.” Cureus, July 2018. doi: 10.7759/cureus.2919.
[5] Gale AH. “Drug Company Compensated Physicians Role in Causing America’s Deadly Opioid Epidemic: When Will We Learn?” Mo Med, July 2016.
[6] “Measles Cases and Outbreaks.” Center for Disease Control, November 2020.
[7] Kramer S. “More Americans say they are regularly wearing masks in stores and other businesses.” Pew Research Center, August 2020.
[8] Zhang K, Vliches TN, Tariq M, Galvani AP, Moghadas SM. “The impact of mask-wearing and shelter-in-place on COVID-19 outbreaks in the United States.” International Journal of Infectious Diseases, December 2020. doi: 10.1016/j.ijid.2020.10.002.
[9] “Frequently Asked Questions for Veklury (remdesivir).” U.S. Food and Drug Administration, October 2020.
[10] COVID-19 Treatment Guidelines Panel. “Coronavirus Disease 2019 (COVID-19) Treatment Guidelines.” National Institutes of Health, Accessed November 2020.
[11] “Researcher Criticizes FDA’s Exaggeration Of Plasma’s Efficacy In COVID-19 Treatment.” All Things Considered. NPR, August 2020.
[12] “Coronavirus (COVID-19) Update: FDA Revokes Emergency Use Authorization for Chloroquine and Hydroxychloroquine.” U.S. Food and Drug Administration, June 2020.
[13] Corum J, Wee S, Zimmer C. “ Coronavirus Vaccine Tracker.” The New York Times, January 2021.
[14] Duke Science & Society. “Coronavirus Conversations: On the Ground – Inside the COVID-19 Vaccine Trials.” Online video clip. Youtube, 6 November 2020.
[15] Duke Science & Society. “Coronavirus Conversations: Emergency Use Authorizations, Public Trust, and Vaccines.” Online video clip. Youtube, 7 October 2020.
[16] “Emergency Use Authorization for Vaccines to Prevent COVID-19; Guidance for Industry.” U.S. Food and Drug Administration, October 2020.
[17] Johnson NF, Velásquez N, Restrepo NJ, Leahy R, Gabriel N, Oud SE, Zheng M, Manrique P, Wuchty S, Yonatan L. “The online competition between pro- and anti-vaccination views.” Nature, May 2020.
[18] Funk C, Kennedy B, Hefferon M. “Vast Majority of Americans Say Benefits of Childhood Vaccines Outweigh Risks.” Pew Research Center, February 2020.