By Neha Madugala, Cognitive Science, ‘22
Author’s Note:
I came across an article detailing the future of surgery. What initially seems like science fiction may be becoming a reality as more and more surgeries are being administered by robots. Through my research, however, I found that robot-assisted surgeries may have the initial appeal of lowering human error, but there are still various issues that must be resolved before they can fully take over in the surgical room.
Robot-assisted surgeries boast the potential of shorter recovery time, less pain and blood, and fewer scars and infections. They have been on the market for a little less than twenty years, and have been used in cancer procedures for about the past fifteen years. While the FDA has approved these devices for other procedures, robot-assisted surgeries have not officially been approved for cancer treatments. Regardless, surgeons have been and continue to perform robot-assisted surgery for cancer-related procedures due to their benefits and increased efficiency.
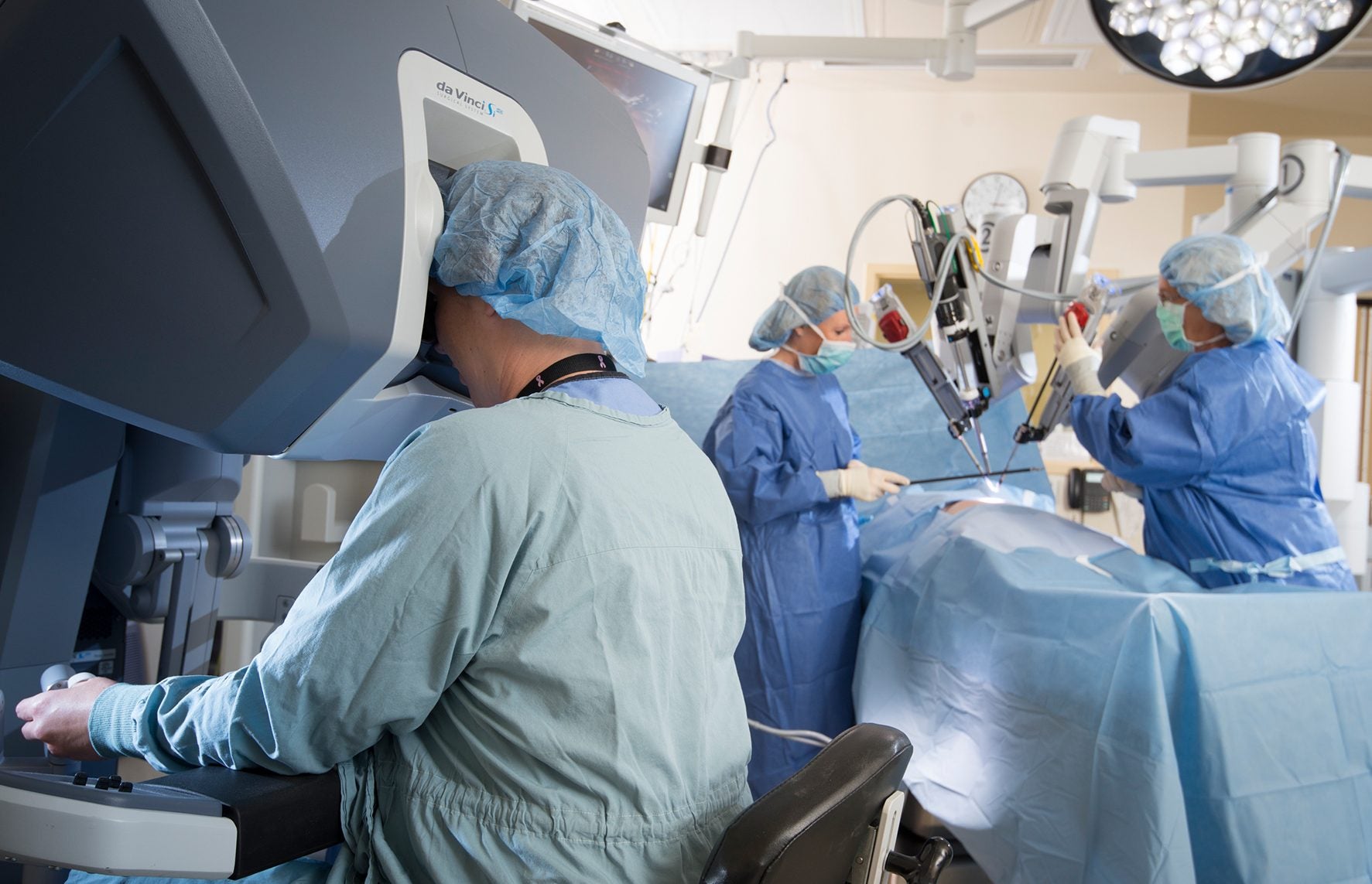
Robot-assisted surgeries mainly contrast from traditional surgeries because they can be performed through small cuts in the patient’s body. As a result, they are minimally invasive. There are three robotic arms, allowing for multiple angles for improved accuracy, which perform the incisions. According to The New York Times, the robotic arms are controlled by a computer and software that replicates the operating surgeon’s movements. This occurs as the operating surgeon performs the movements while looking at a magnified and high-definition screen of the surgical site captured by a camera attached to the robot. While the device requires limited retraining for surgeons, as of now, there is only one company actually offering this device. Interestingly, the device requires less precision and attention by the surgeon due to the magnification and the actual incisions being performed by the robot.
In 2000, the FDA approved for one of the first robot-assisted systems to be brought to the market. The system, called the da Vinci Surgical System promised to improve the efficiency and effectiveness of medical surgeries, not just cancer-related surgeries. In order to bring the system to the market quickly, the robotic surgery system went through “premarket notification,” allowing the company to skip the rigorous safety and efficacy trials. Essentially, “premarket notification” is supposed to ensure that a device is safe and this notation helps quicken a device’s journey to the market. The FDA said that this decision was based only on short-term data and a spokesperson stated that the decision was made “based on evaluation of the device as a surgical tool and did not include evaluation of outcomes related to the treatment of cancer.” The device promises more successful surgeries with limited retraining and a smooth transition from a humancentric to robot-assisted surgery. These prospects posed limited risks and the evident benefit of improving the success rate of these surgeries; as a result, the device was approved without a thorough and holistic evaluation.
While this system has only been approved for some urological and gynecological procedures, these devices are used for a vast array of other unapproved procedures. The FDA can assess the safety of these devices for certain procedures, but they cannot prevent these systems from being used in unapproved settings in the medical field. As a result, medical professionals may still use these systems for procedures that have not been approved by the FDA.
At the beginning of March, the FDA released a statement reminding the public that robot-assisted surgeries have not been approved for mastectomy or cancer-related surgeries, two procedures for which the device is frequently used. Dr. Terri Cornelison, who works for the FDA’s Center for Devices and Radiological Health, has stated, “We are warning patients and providers that the use of robotically-assisted surgical devices for any cancer-related surgery has not been granted marketing authorization by the agency. The survival benefits to patients when compared to traditional surgery have not been established.” The FDA has claimed that there is no supporting evidence that robot-assisted surgeries are better than traditional surgeries and they have further claimed that robot-assisted surgeries result in more problems for patients receiving treatment for cervical cancer. Cornelison further states, “We want doctors and patients to be aware of the lack of evidence of safety and effectiveness for these uses so they can make better informed decisions about their cancer treatment and care.”
The FDA cited two studies that warn against the danger of robot-assisted surgery. Both studies were published by the New England Journal of Medicine. Both studies analyzed the difference between robot-assisted and traditional procedures for cervical cancer in women. The first study found that women who received surgery with robotic methods faced four times as many cancer recurrences and six times as many deaths. It should be noted that the procedure – radical hysterectomy – is considered to be a relatively safe procedure when performed correctly that can cure patients of cervical cancer. Furthermore, in the second study, 9.1% of the sample group died after minimally invasive surgeries, or in other words robot-assisted surgeries, and 5.3% died in open surgeries, which involve no robotic mechanisms.
It is not clear why robot-assisted surgeries have had worse results for cervical cancers. Dr. Pedro T. Ramirez, a surgical researcher at the Anderson Cancer Center in Houston, believes that these results may be due to the device or because carbon dioxide, which is used to provide a working and viewing space for the surgeon, may increase the spread of cancer during the procedure.
These findings by the FDA encourage patients to question their medical professionals about what type of procedures they will receive and to know the facts about different methods for surgery. In order to ensure that they receive the best care, it is important that patients have a say in the procedure they will receive by accurately weighing the risks and benefits. While the FDA cannot stop the use of these tools in the medical field, increased interest and probing of the mechanics of these systems are helping raise awareness about what is actually happening in the operation room.